 For example, CdnC from Escherichia coli ( EcCdnC) and CdnD from Pseudomonas aeruginosa ( PaCdnD) synthesize cyclic tri-AMP (cAAA) as a component of cyclic oligonucleotide-based anti-phage signaling system (CBASS) ( 14). Recently, scores of newly discovered cGAS/DncV-like nucleotidyltransferases (CD-NTases) were shown to produce a variety of CDNs and cyclic trinucleotides (CTNs) ( 13). The nucleophilic attack of O3′ (or O2′) at α-phosphate is indicated by an arrow. The Mg 2+ ion bound to the acceptor ribose is designated metal A, and that bound to the donor triphosphate is metal B. 
The shared mechanism of NTase-catalyzed reaction that forms CDN or CTN is depicted in ( D). Hydrogen atoms and some double bonds are not shown. The chemical structures of three cyclic nucleotides are presented as schematic diagrams in ( A) cAMP, ( B) 2′,3′-cGAMP and ( C) cAAG. These enzymes all belong to the SMODS (second messenger oligonucleotide or dinucleotide synthase) family of proteins ( 12).Ĭyclic nucleotides. It binds to a CDN-activated phospholipase ( VcCapV), which degrades the bacterial cell membrane ( 11). 3′,3′-cGAMP is synthesized by dinucleotide cyclase in Vibrio cholerae ( VcDncV), a cGAS homologue ( 10). STING, serving as a pattern recognition receptor (PRR), also recognizes the bacterial CDNs as pathogen associated molecular patterns (PAMPs), which include 3′,3′-cGAMP in addition to c-di-GMP and c-di-AMP ( 9). Innate immune responses are triggered by 2′,3′-cGAMP binding to STING (stimulator of interferon genes) and by 2′,5′-oligoadenylate activation of RNase L ( 7, 8). In mammals, 2′,3′-cyclic GMP-AMP (2′,3′-cGAMP Figure 1B) with the guanosine C2′ forming a non-canonical phosphodiester bond to the adenosine C5′, and 2′,5′-oligoadenylate with similar linkages, are synthesized by cGAS and OAS upon activation by double-strand DNA (dsDNA) and dsRNA ( 5, 6). Many bacterial cell processes are regulated also by 3′,5′-linked cyclic dinucleotides (CDNs) such as c-di-GMP and c-di-AMP ( 3, 4). In bacteria, cAMP activates genes for catabolic pathways ( 2). For example, 3′,5′-cyclic adenosine monophosphate (cAMP Figure 1A) promotes glycogen breakdown in human ( 1). Based on structural analysis and comparison with VcDncV and RNA polymerase, a tentative catalytic pathway for the CTN-producing EcCdnD is proposed.Ĭyclic nucleotides have long been known to serve as second messengers that regulate cellular activities in response to environmental variations. The conserved residues Asp69 and Asp71 are essential for catalysis, as indicated by the loss of activity in the mutants. The enzyme is active on ATP or a mixture of ATP and GTP, and the best metal cofactor is Mg 2+. GTP alone does not bind to EcCdnD, which however binds to pppApG, a possible intermediate. Isothermal titration calorimetry results also suggest the presence of two ATP binding sites. Adjacent to the donor substrate, another nucleotide is bound to the acceptor binding site by a non-productive mode. Despite the similar overall architecture, the protein shows significant structural variations from other CD-NTases. Here, we present the crystal structures of EcCdnD, a CD-NTase from Enterobacter cloacae that produces cyclic AMP-AMP-GMP, in its apo-form and in complex with ATP, ADP and AMPcPP, an ATP analogue. Recently, scores of new cGAS/DncV-like nucleotidyltransferases (CD-NTases) were discovered, which produce various CDNs and cyclic trinucleotides (CTNs) as second messengers. 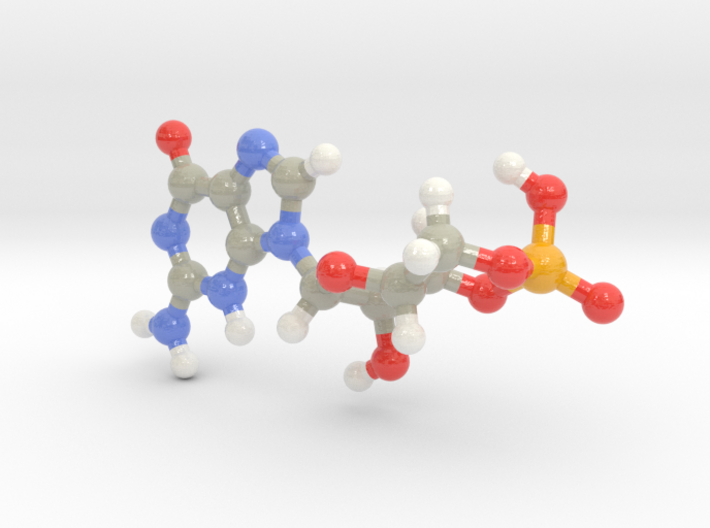
These results indicate an imbalance in cyclic nucleotides metabolism in psoriatic skin without a significant difference between uninvolved and involved epidermis.Mammalian cyclic GMP-AMP synthase (cGAS) and its homologue dinucleotide cyclase in Vibrio cholerae ( VcDncV) produce cyclic dinucleotides (CDNs) that participate in the defense against viral infection. Thus, the ratio cAMP/cGMP was higher in normal skin than in psoriatic skin at the basic level (p < 0.05) and after incubation (p < 0.01). 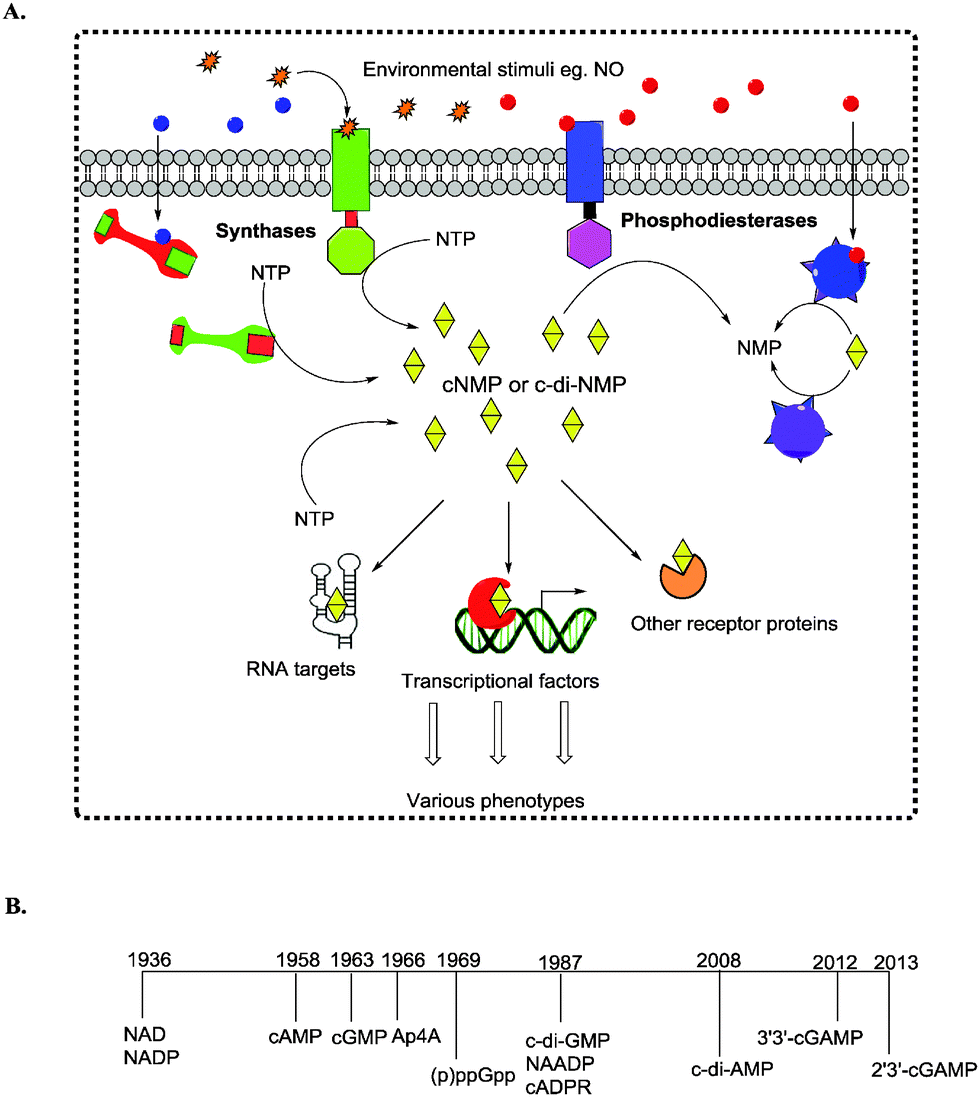
After incubation, psoriatic involved epidermis exhibited a higher production of cGMP than normal skin (p < 0.05). cGMP levels were not significantly different in the three tissues at zero time. Comparison of normal skin, uninvolved and involved psoriatic skin, prior to and after 20 min of incubation showed the following results: cAMP levels were significantly higher in normal skin than in psoriatic skin at zero time (p < 0.01) and after incubation (p < 0.05). The kinetics of cAMP and cGMP production were linear during the 20 min of incubation. Cyclic nucleotides were measured by radioimmunoassay before and after 5, 10,20 min of incubation. After homogenization, epidermal strips were incubated with saturating concentrations of ATP or GTP, respectively, for cAMP and cGMP production. CAMP and cGMP production was determined in normal and psoriatic (involved or uninvolved) epidermis. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
